The Reduction of Traumatic Spinal Cord Secondary Injury by Anti-RhoA siRNA Functionalized Nucleic Acid Nanoparticles (NANPs)
Original Research Article
DOI:
https://doi.org/10.59566/isrnn.2024.0101079Keywords:
Spinal cord injury, NANPs, RhoA/Rho kinase, RNAiAbstract
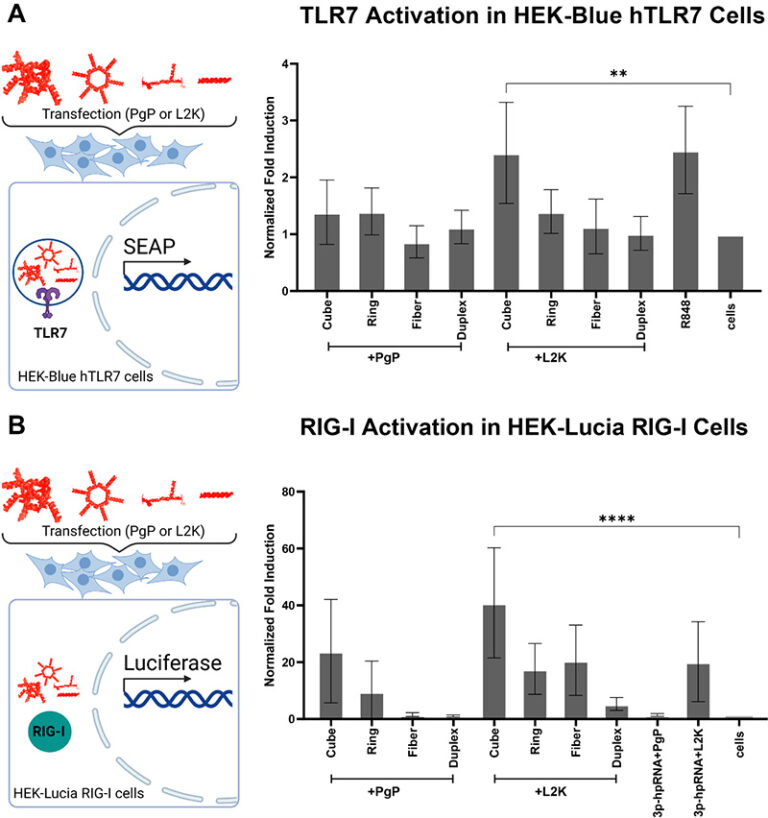
Primary injury of the spinal cord is caused by a mechanical traumatic event which is rapidly followed by a secondary injury cascade that may evolve for several months leading to biological and functional changes. During the secondary injury, many pathophysiological pathways and process are activated including inflammation, oxidative stress, demyelination, excitotoxicity, axon degeneration, and cell death. The RhoA/Rho kinase pathway significantly contributes to spinal degeneration and regeneration and therefore represents a potential therapeutic target. Nucleic acid nanoparticles (NANPs) offer easy rational and programable design with the potential to carry on multiple synergistic therapeutic nucleic acid functional motifs. In this context we designed, synthesized, and assembled several representative NANPs decorated with multiple copies of siRNAs targeting RhoA. Subsequently we assessed NANPs' physicochemical properties, toxicity, and immunorecognition upon delivery with the nanocarrier PLGA-g-PEI (PgP), developed with the aim to select for the most immunoquiescent type of formulations. In addition, we observed that L1 neural cell adhesion molecule conjugated PgP (L1-PgP) efficiently delivered NANP-siRhoA in cultured neuroblastoma (B35) cells. RhoA mRNA expression was significantly reduced by all L1-PgP/ NANP-siRhoA relative to the untreated control, while no significant differences were observed between the different NANP-siRhoAs.