Co-incubation of Short Amphiphilic Peptides with Dicer Substrate RNAs Results in β-Sheet Fibrils for Enhanced Gene Silencing in Cancer Cells
Original Research Article
DOI:
https://doi.org/10.59566/isrnn.2024.0101061Keywords:
amphiphilic peptides, β-sheet fibrils, RNA interference, Dicer substrate RNA, RNA-peptide co-evolution, peptide folding energy landscape, cancerAbstract
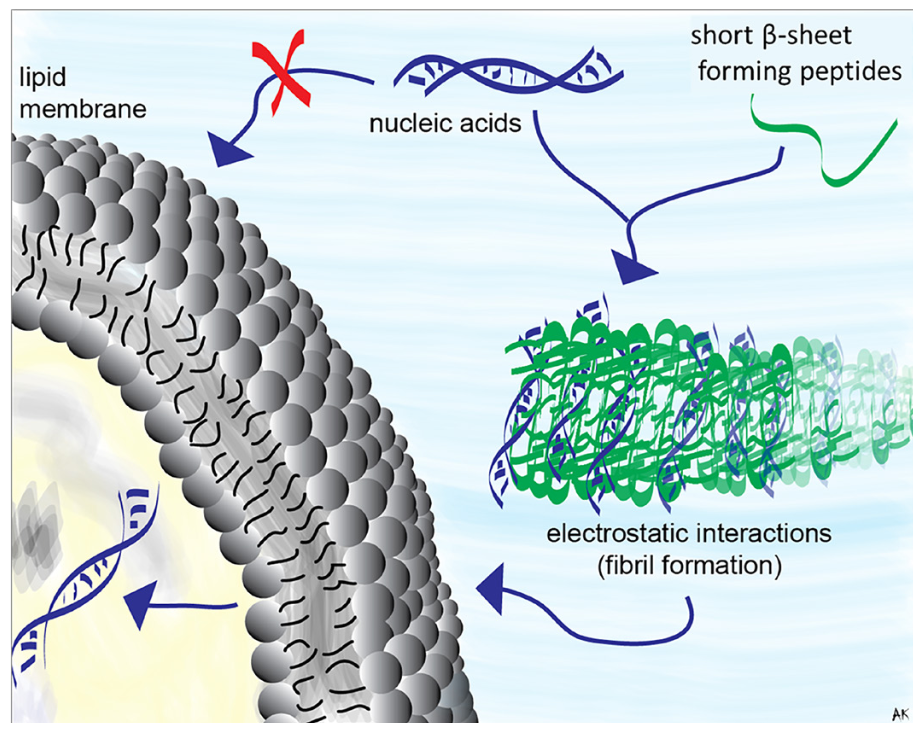
RNA can interact with positively charged, amphiphilic peptides to cooperatively assemble into fibrils that enable RNA transport across cancer cellular membranes. RNA decreases the folding energy barrier imposed by the electrostatic repulsion between these charged peptides, thus partaking in RNA-peptide self-assembly along particular pathways in the energy landscape. Specific amphiphilic peptides capable of protecting and transporting RNA across a membrane have Type II’ β-turn hairpin forming motifs in their structures, which aids self-assembly into β-sheet fibrils. We employed a set of such cationic, amphiphilic peptides that have random coiled structures in the absence of folding stimuli, to characterize the (peptides):(RNA) assembly. We subjected these complexes to extensive biophysical characterization in vitro and in cell culture. We show that short RNAs (such as Dicer substrate RNAs) can lead these peptides to self-assemble into β-sheet fibrils that have RNA transport capabilities and can act as non-viral delivery vectors for RNA. Modulation in the peptide sequence implicitly alters the way they bind RNA and influence the peptides’ ability to transport nucleic acids across membranes.