Conversion of Chemical Drugs into Targeting Ligands on RNA Nanoparticles and Assessing Payload Stoichiometry for Optimal Biodistribution in Cancer Treatment
DOI:
https://doi.org/10.59566/isrnn.2024.0101109Keywords:
Methotrexate, RNA nanoparticles, three-way junction (3WJ), click reaction, cancer targetingAbstract
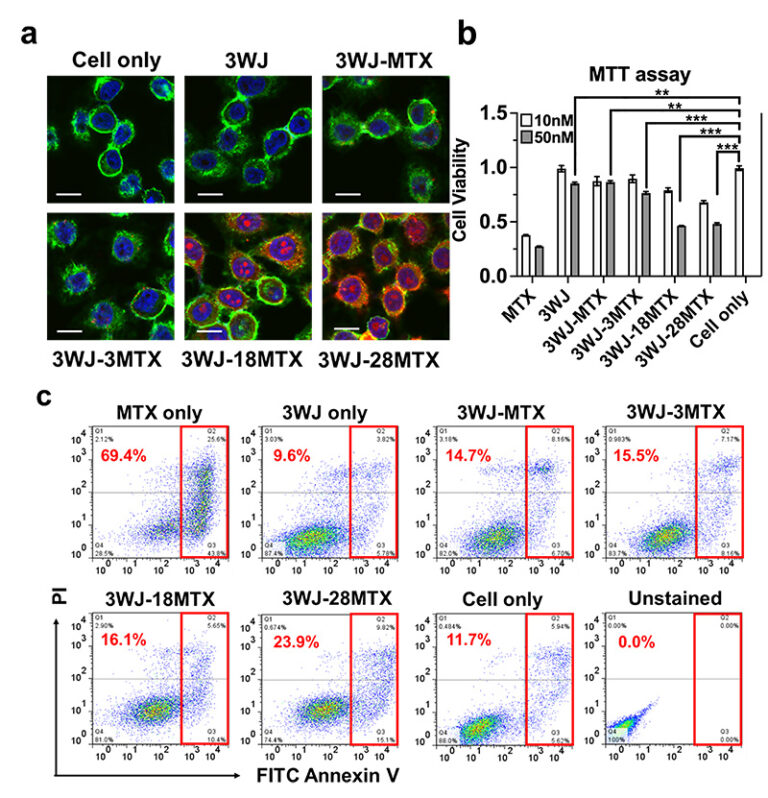
Active targeting-mediated nanodelivery takes advantages of ligand-receptor specificity to avoid non-specific distribution, holding great promise for the treatment of a spectrum of diseases. RNA nanoparticles have demonstrated rapid spontaneous tumor targeting and very little organ accumulation due to rapid renal clearance of non-tumor accumulated RNA nanoparticles. However available ligands for specific cells are limited, yet many chemical entities possess receptor targeting capability and remains unexplored. To provide specific tumor accumulation, a multivalent targeting strategy on RNA nanoparticles to control their in vivo fate is implemented. Methotrexate (MTX), a clinically approved chemotherapy was used as a tumor-targeting ligand through conjugation to our RNA nanoparticle with controlled conjugation of various copy numbers. As copies of conjugated MTX increased on the nanoparticle, the specific binding to overexpressed folate receptor was enhanced as demonstrated by flow cytometry analysis and confocal microscopy imaging. Increasing the amounts of conjugated MTX did not significantly change the nanoparticle size, Zeta potential, or cytokine induction. Increased amounts of conjugated MTX resulted in improved cell inhibition due to MTX release following cell internalization. However, increasing conjugated MTX to the RNA nanoparticles reduced the melting temperature of RNA nanoparticles and increased in vitro serum protein binding to the nanoparticles. Thus, in vivo biodistribution profiles of RNA nanoparticles revealed different behaviors based on MTX conjugation in cancer targeting and clearance. Increased copies of MTX changed the ability of nanoparticles to target tumors, accumulate in healthy organs, and rapidly clear through the urine. Nanoparticle design must be closely considered for optimized cancer targeting and therapy, providing the rationale for a proper design of RNA nanodelivery in cancer treatment.